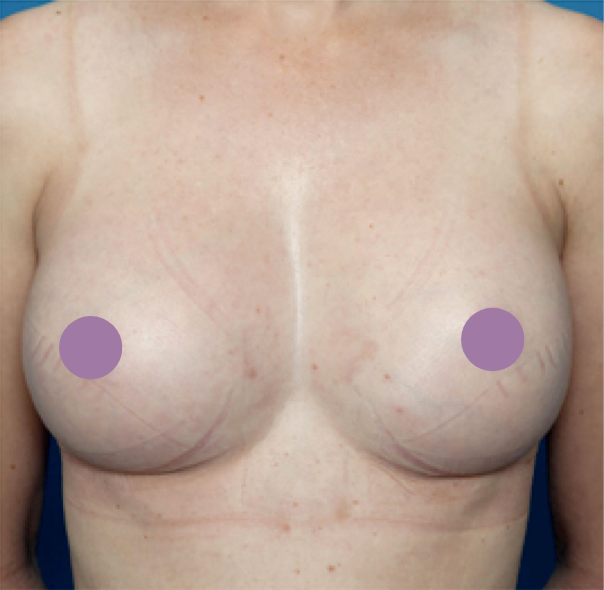
We have extensive experience in performing Breast Implant Revision Surgery for a variety of cases.
Often the wrong type of implant or the wrong shape implants have been used, or the implants are too big, too small. We have found that with Overseas Surgery for Breast Augmentation Surgery generally a “production line” approach is used where the same type and size of implants are used on each patient, generally with poor results.
In some patients the breast implants have “encapsulated” where the patient’s body has rejected the implants which require the original implant (or implants) to be removed and replaced.
Surgical Warning: Any surgical or invasive procedure carries risks. Before proceeding you should seek a second opinion from an appropriately qualified health practitioner.
Read more on the Risks of Surgery
What is Breast Implant Revision surgery?
Implant Revision Surgery is the replacement of your existing breast implants and it involves a no obligation consultation with one of our Surgeons who you can discuss your concerns and problems.
Our Surgeons first understand your problem, your requirements and your expectations and then make recommendations to ensure that your Implant Revision surgery fulfils your expectations.
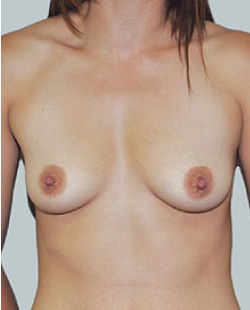
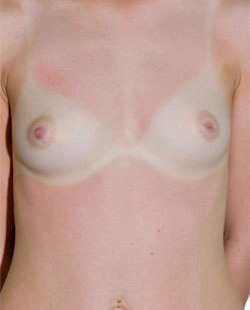
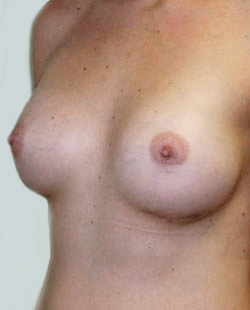
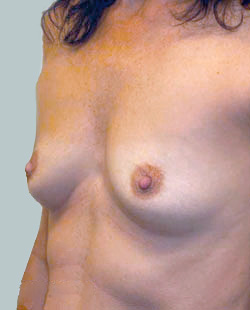
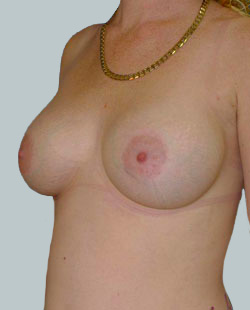
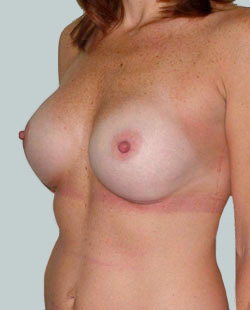
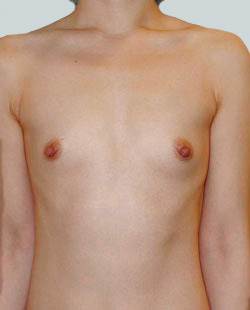
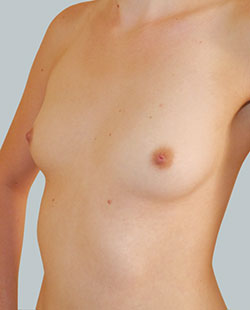
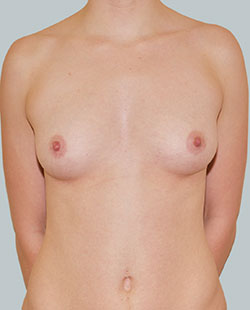
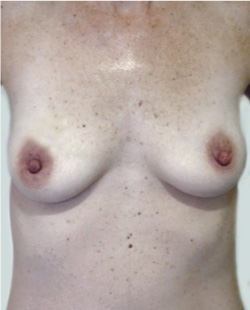
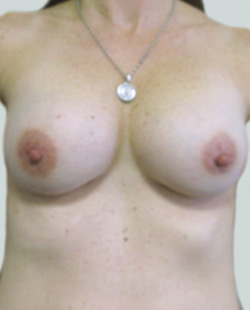
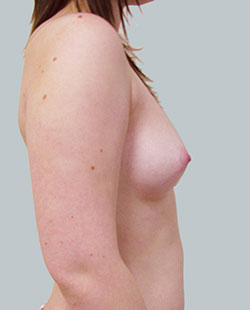
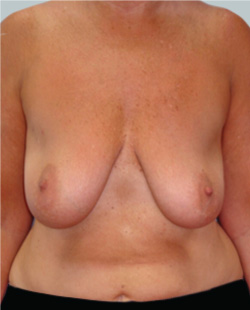
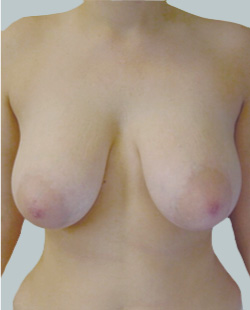
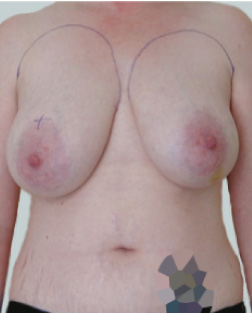
Before 
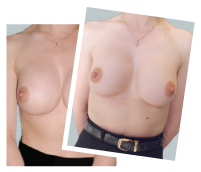
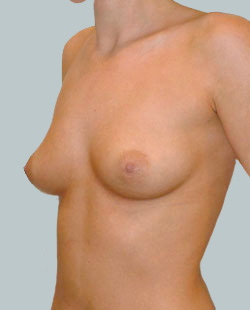
This 43-year-old female had previous breast augmentation and wished to seek consultation on further augmentation to alter the look of their chest area.
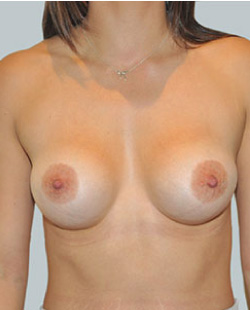
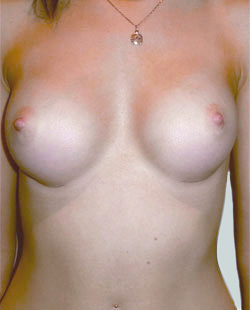
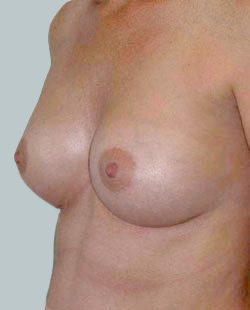
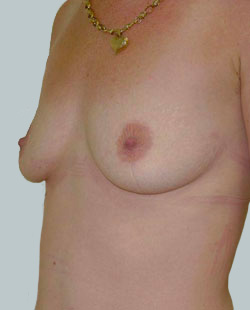
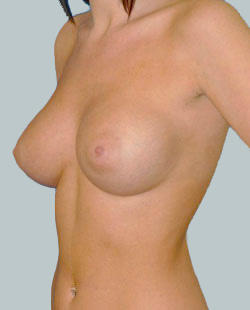
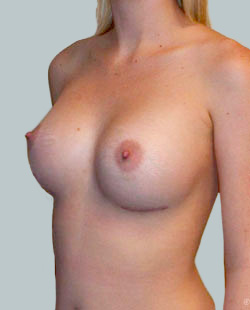
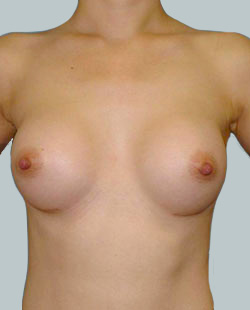
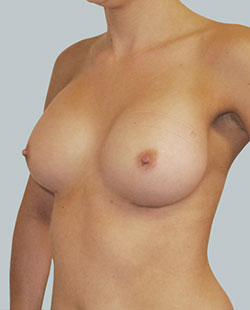
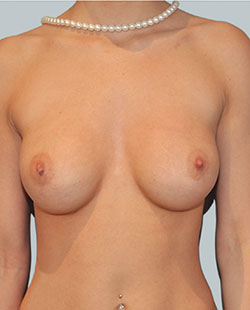
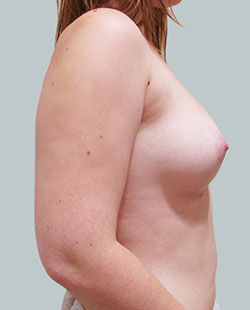
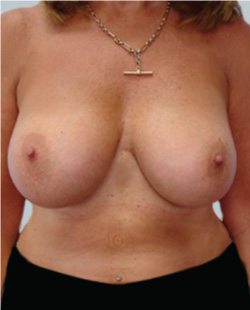
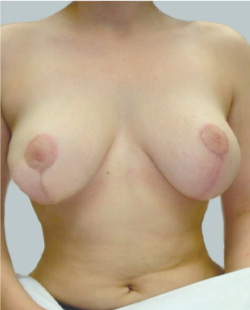
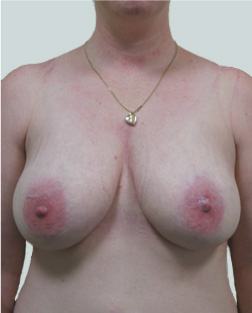
After 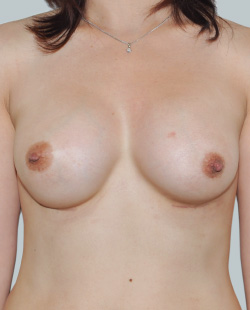
Breast Augmentation was performed, using two different-sized round implants to correct and even out the breast area.
Performed By: Dr Simon Rosenbaum (MED0000957217)
What should I expect after Implant Revision Surgery?
After your procedure, you may experience some discomfort and drowsiness due to the anaesthetic. It is important to have someone drive you home and stay with you for at least the first night. Rest is crucial in the initial stages of recovery. However, full recovery may take several weeks and may include swelling, bruising, and restricted activity. We will provide detailed post-operative care instructions to aid your recovery. You are provided with an after hours direct number in the event you may have any questions or concerns you wish to speak to us about.
The next day or within a few days you must come back to the Clinic for your first post-operative check.
After some initial swelling and tightness over the first few days after your procedure, you should start to see the shape of your breasts or the new shape if different size or shape implants have been used by our Surgeons. As with your initial surgery you would be aware that further changes may occur over the next few months as your body adjusts to the implants.
We recommend avoiding strenuous activity for around three weeks, but from six weeks or so you will be able to return to any exercise routines you had before. Our Surgeons and Nurses will continue to monitor your recovery through a number of post-operative check-ups and are available at any time if you have questions or concerns.
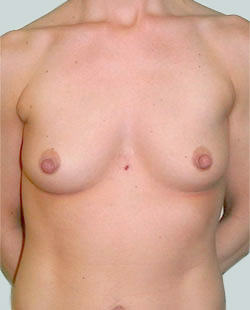
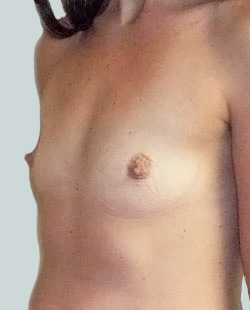
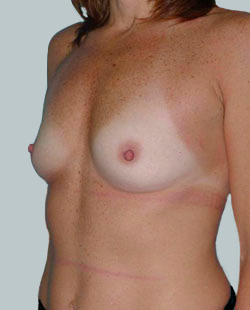
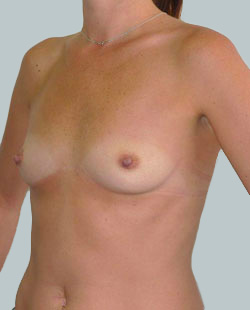
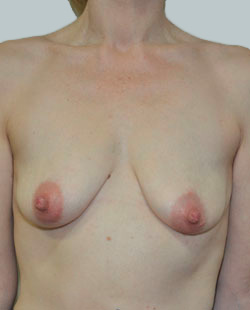
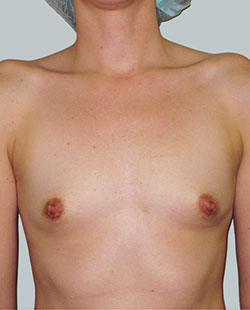
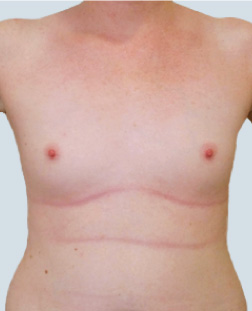
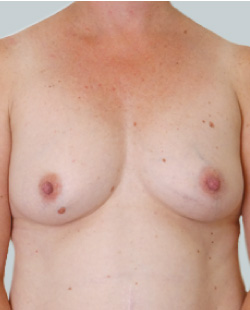
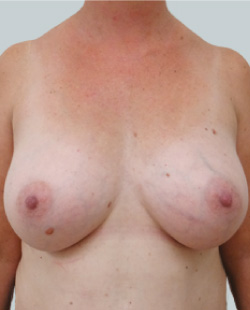
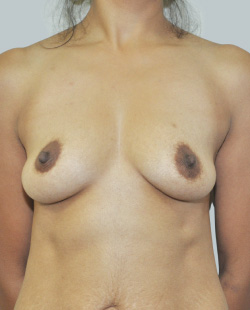
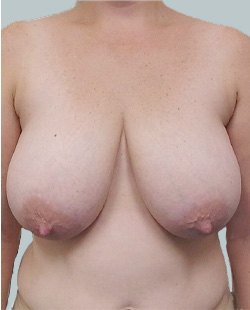
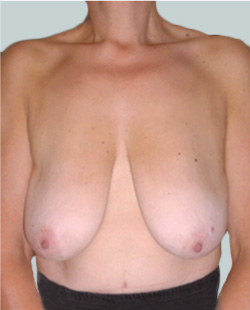
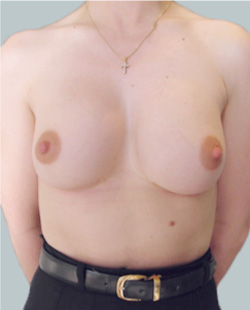
Before 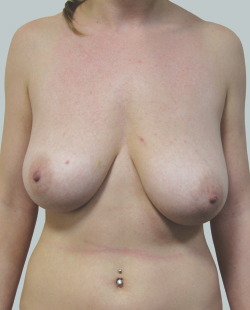
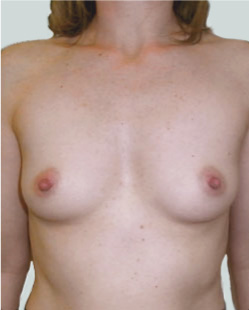
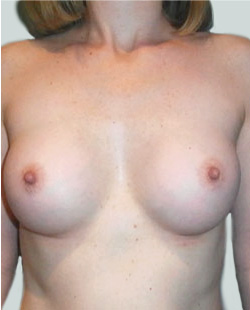
This 24-year-old patient sought advice on breast surgery to reduce the size of and reshape their chest area.
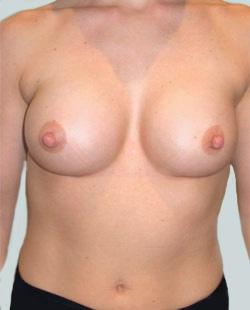
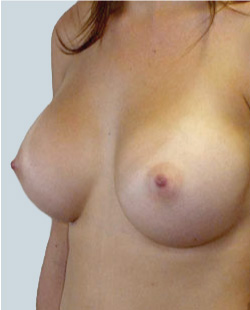
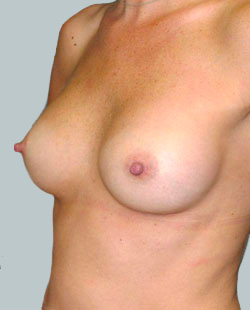
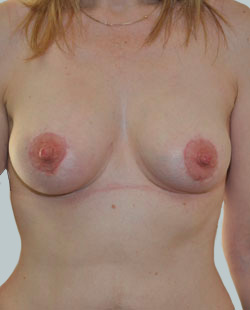
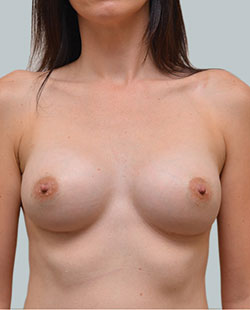
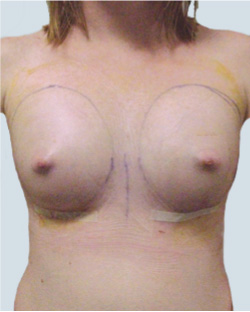
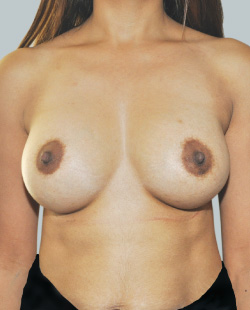
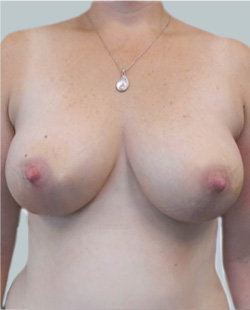
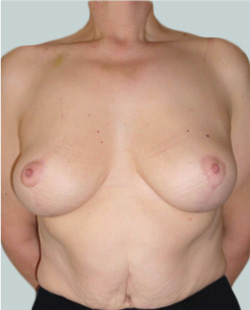
After 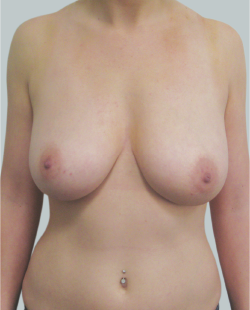
Liposuction to the breast was performed to reduce the size of the breast and make the breasts more symmetrical.
Performed By: Dr Ashley Granot (M0001026218) MBBS, FFMACCS (Med), FACP, FACNEM, ABBRM, MA5M Cosmetic Doctor, Melbourne
How Much Does Implant Revision Cost?
The cost of Implant Revision Surgery can vary based on individual circumstances. This includes the cost of consultations, the surgery itself, anaesthesia, and any necessary aftercare. We will provide a detailed cost breakdown during your consultation. A no obligation consultation with one of surgeons at the Me Clinic is essential in determining what you require and which procedure would be best suited for you.
Our philosophy is simple. We want you to be fully informed, and we want you to understand all your options and have one accurate price quoted once. Consultation fees may apply.
There is no need for you to obtain a referral from your own doctor to see one of our Surgeons. Generally we are able to book you in to see one of our Surgeons in 7 to 14 days.
How do I find out more about Implant Revision Surgery?
Please contact the Me Clinic. We have over 35 years of Cosmetic Surgery experience and we have performed over 5,000 successful Breast Augmentation procedures. We ensure you will be fully informed and understand your options before any procedure.
Surgical Warning: Any surgical or invasive procedure carries risks. Before proceeding you should seek a second opinion from an appropriately qualified health practitioner.
What are the risks of breast implant revision surgery?
Common risks of every surgical procedure include:
- Infection
- Bleeding or hematoma
- Scarring
- Adverse reactions to anesthesia
- Blood clots
- Pain and discomfort
- Swelling and bruising
- Potential need for revision surgery
Implant specific complications include:
- Capsular contracture
- Implant displacement
- Rupture or leakage
- Rippling or wrinkling
- Breast pain
- Breast implant-associated anaplastic large cell lymphoma (BIA-ALCL)
- Breast implant illness (BII)